Critical Needs in Cellular Target Engagement
Cellular Target Engagement Challenges and Needs
The development of new medicines for treating diseases and new chemical probes for elucidating biological function continues to rely on new or improved small molecules that engage a protein target. Additionally, confirmation of the drug’s mode of action in living cells remains a critical step for chemical biology and pharmacological research.
Demonstration of cellular target engagement is an essential step for confirming a compound’s mechanism of action as it can verify that a drug reaches the intended organ or tissue, is cell-penetrant, and engages a specific target, all in a manner consistent with an observed phenotypic outcome. Thorough characterization of cellular drug binding can accelerate drug discovery and ultimately reduces clinical failure rates and associated high costs to drug development.
Cellular Target Engagement Methods
Monitoring compound-target engagement in the cellular milieu is essential to validate the pharmacology of drug candidates. Current cellular target engagement methods vary in assay read-out types, include labelling to label-free techniques, and range from easy to complex data evaluation.
Compound-Target Engagement Cellular Methods
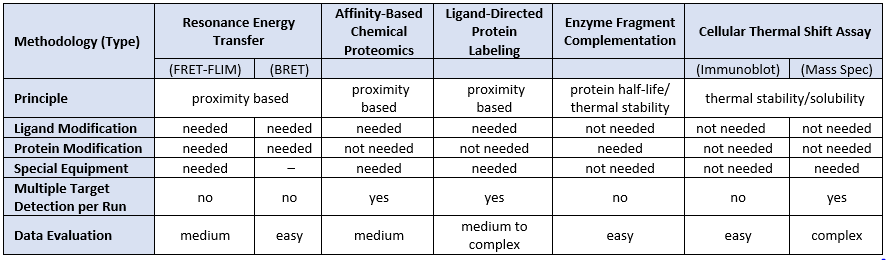
Modified from Schϋrmann et al., 2016
Enzyme Fragment Complementation Method – InCELL Assays
DiscoverX’s proprietary enzyme fragment complementation (EFC) technology is based on two recombinant fragments of a β-galactosidase and a chemiluminescent read-out. The technology is simple and flexible – can be applied to study not only compound-target engagement, but additionally protein-protein interactions, degradation, activity and functional characterization, and more.
For cellular compound-target engagement, EFC is utilized to analyze the interaction based on either a change in protein half-life (InCELL Hunter™) or protein thermal stability (InCELL Pulse™). These two related EFC approaches together increase the applicability of the assays and the number of protein targets covered. These simple InCELL assays require no ligand modification or antibody reagents and provide a convenient solution when functional assays are difficult or unavailable. Additionally, the assays are validated and designed for screening inhibitors, validating hits identified in biochemical assays, confirming compound cell entry, measuring cellular EC50 values, and ranking compounds during lead optimization – all in the native cellular environment.
Overall, InCELL assays have been successfully applied to several cytosolic and nuclear proteins such as kinases, methyltransferases, and hydrolases, and they provide the most flexible solution in the form of products (cell lines, ready-to-use kits, and do-it-yourself products) and services (screening services or custom assay development).
InCELL Compound-Target Engagement Assay Highlights
- Easily measure compound binding to your protein target with a chemiluminescent readout
- Rapidly make your own target engagement cell-based assays in <2 weeks
- Generate biologically relevant data for more confident drug discovery decisions
To learn more about InCELL Target Engagement visit discoverx.com/incell.
InCELL Target Engagement Resources and References
Application Notes:
- InCELL Pulse: Cellular Target Engagement Assays for Characterizing Novel Small Molecule Inhibitors of Oncogenic SHP2 Variants
- InCELL Hunter: Automating a Cell-Based Target-Compound Assay for Methyltransferase and Bromodomain Proteins using the InCELL Hunter Platform
Posters
- InCELL Pulse: A Novel Cellular Target Engagement Assay Platform for Drug Discovery
- Treiber, et al. DiscoverX (SLAS 2017, DOT 2016)
- Reviving up CETSA® (this poster uses the InCELL Pulse technology)
- McNully, et al. GSK (SLAS 2016)
- CETSA is a registered trademark of Pelago Biosciences
- Utilisation of the InCELL Hunter Target Engagement Cell-Based Assay for Profiling of ULK1 Inhibitors
- Newman, et al. MRC (ELRIG 2015)
- Evaluation of InCELL Hunter Technology for use with Bromodomain targets
- Gatfield and Craggs GSK (SLAS 2015)
Publications
- A High-Throughput Dose-Response Cellular Thermal Shift Assay for Rapid Screening Drug Target Engagement in Living Cells, Exemplified Using SMYD3 and IDO1
- McNulty DE, et al. SLAS Discov. 2018 Jan; 23(1): 34-46. PMID:28957646
- Small-Molecule Target Engagement in Cells (minireview)
- Schϋrmann, et al., Cell Chem Biol. 2016, 23(4) 435-41.
- Examining Ligand-Based Stabilization of Proteins in Cells with MEK1 Kinase Inhibitors
- Auld DS, et al., Assay Drug Dev Technol. 2015, 13(5) 266-76.
- Cell-Based Protein Stabilization Assays for the Detection of Interactions between Small-Molecule Inhibitors and BRD4
- Schulze et al., J. Biomol. Screening. 2015, 20(2) 180-89.
- A Potent, Selective and Cell-Active Allosteric Inhibitor of Protein Arginine Methyltransferase 3 (PRMT3)
- Kaniskan et al., Angew Chem Int Ed Engl. 2015, 45(17) 5166-70.
